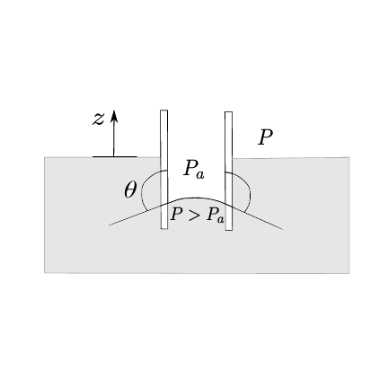
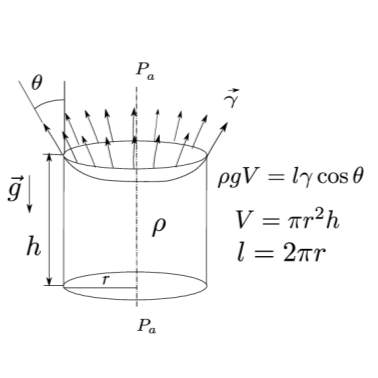
2.3.1. Exercise 2.1#
Let \(\theta\) be the contact angle at the interface between air, liquid, and solid;
let \(\gamma\) be the surface tension between air and liquid; let \(\rho\) be the density of the liquid.
Determine the height \(h\) of the liquid in a cylindrical capillary tube of radius \(r = 0.5 \ mm\) relative to the level in the reservoir.
Then calculate the pressure inside the column.
(We can assume the approximation that the pressure acting on the reservoir and on the top surface of the liquid in the capillary is equal.)
Assume thermodynamic conditions and tube material such that:
if the liquid is water: \(\rho = 999 \ kg/m^3\), \(\theta={1}^\circ\), \(\gamma=0.073 \ N/m\)
if the liquid is mercury: \(\rho = 13579 \ kg/m^3\), \(\theta={140}^\circ\), \(\gamma=0.559 \ N/m\)
(\(h_{H_2O} = 2.97 \ cm\), \(P_{H_2O} - P_0 = - 291.95 \ Pa\); \(h_{Hg} = -1.28 \ cm\), \(P_{Hg} - P_0 = 1712.87 \ Pa\))
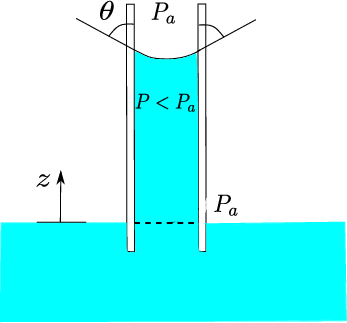
Concepts. Surface tension. Contact angle. Capillarity. Meniscus.
Solution. By writing the equilibrium for the volume of fluid in the capillary, the height \(h\) is obtained. Then the pressure \(p\) is found using Stevin’s law. Finally, observations are made about the contact angle, meniscus, and pressure jump across the interface.
Write the equilibrium for the fluid volume. This is a statics problem.The acting forces are the surface tension (acting on the perimeter of the top surface) and the weight force, since by assumption the pressure acting on the top surface equals ambient pressure \(P_a\) (So??? Why is the vertical component of the external pressure resultant zero??? See figure below).
\[F_{\gamma} = F_g \quad \Rightarrow \quad 2\pi r \gamma \cos \theta = \pi r^2 h \rho g\]And therefore:
\[\begin{split}h = \frac{2 \gamma \cos \theta}{\rho g r} \quad \Rightarrow \quad \begin{cases} h_{H_2O} = 2.97 \ cm \\ h_{Hg} = -1.28 \ cm \end{cases}\end{split}\]Comments on the result.
The capillarity effect is more significant for narrow tubes (inverse proportionality with \(1/r\)). The height \(h\) can be either positive or negative depending on the contact angle: \(h \le 0\) for \(\theta \ge \pi/2\).Calculate the pressure in the fluid at the top of the column using Stevin’s law:
\[\begin{split}P = P_0 - \rho g h = P_0 - \frac{2 \gamma \cos \theta}{r} \quad \Rightarrow \quad \begin{cases} P_{H_2O} - P_0 = - 291.95 \ Pa \\ P_{Hg} - P_0 = 1712.87 \ Pa \end{cases}\end{split}\]Comments on the result. These results are consistent (thankfully!) with the relationships between curvature (closely related to the meniscus and contact angle) and the pressure jump. See pictures below for a comparison of the experiment using either water (\(\theta <\pi/2\)) or mercury (\(\theta > \pi/2\))
water
\(\theta \le \pi/2\)
\(h \ge 0\)
\(P \le P_a\)
mercury
\(\theta \ge \pi/2\)
\(h \le 0\)
\(P \ge P_a\)
|
|
|